 
Multiply the number of calcium atoms by 3 and the number of phosphorus atoms by 2. Phosphorus goes from 0 to -3, gaining 3 electrons (oxidation).Calcium goes from 0 to +2, losing 2 electrons (reduction).Identify the changes in oxidation numbers:.In Ca 3P 2, calcium has an oxidation number of +2, and phosphorus has an oxidation number of -3.It’s important because in a chemical reaction, the quantity of each element does not change (the law of conservation of mass). This means looking for stoichiometric coefficients for the reactants and products. Phosphorus (P) also has an oxidation number of 0 in its elemental form. So, the first step in chemical reaction calculations is balancing chemical equations.Calcium (Ca) has an oxidation number of 0 in its elemental form.Process: identify the oxidation numbers, determine the changes in oxidation state, balance the atoms that change their oxidation state, and then balance the remaining atoms and charges. Useful for redox reactions, this method involves balancing the equation based on the change in oxidation numbers.īest For: Redox reactions where electron transfer occurs. Balance the oxygen atoms by placing a coefficient of 2 in front of H 2O:. 
There are 2 O atoms on the left and 1 O atom on the right. There are 2 H atoms on the left and 2 H atom on the right. Count the number of H and O atoms on both sides.Process: Start with the most complex molecule or the one with the most elements, and adjust the coefficients of the reactants and products until the equation is balanced. It involves looking at the equation and adjusting the coefficients to get the same number of each type of atom on both sides of the equation.īest for: Simple equations with a small number of atoms. Once balanced, it ensures that the atoms for each element will remain equal on both sides of the chemical equation. Find more Chemistry widgets in WolframAlpha. Get the free 'Chemical Equation Balancer' widget for your website, blog, Wordpress, Blogger, or iGoogle. Balancing with inspection or trial and error method Balancing chemical equations calculator automatically balances the chemical equations by adjusting the coefficients of reactants and products to ensure the conservation of atoms. Just enter in each molecule name and it will give the right numbers of each required to balance the equation. A balanced equation obeys the Law of Conservation of Mass, which states that matter is neither created nor destroyed in a chemical reaction. However, this equation isn't balanced because the number of atoms for each element is not the same on both sides of the equation. For example, in the reaction of hydrogen (H₂) with oxygen (O₂) to form water (H₂O), the chemical equation is: It shows the reactants (substances that start a reaction) and products (substances formed by the reaction). 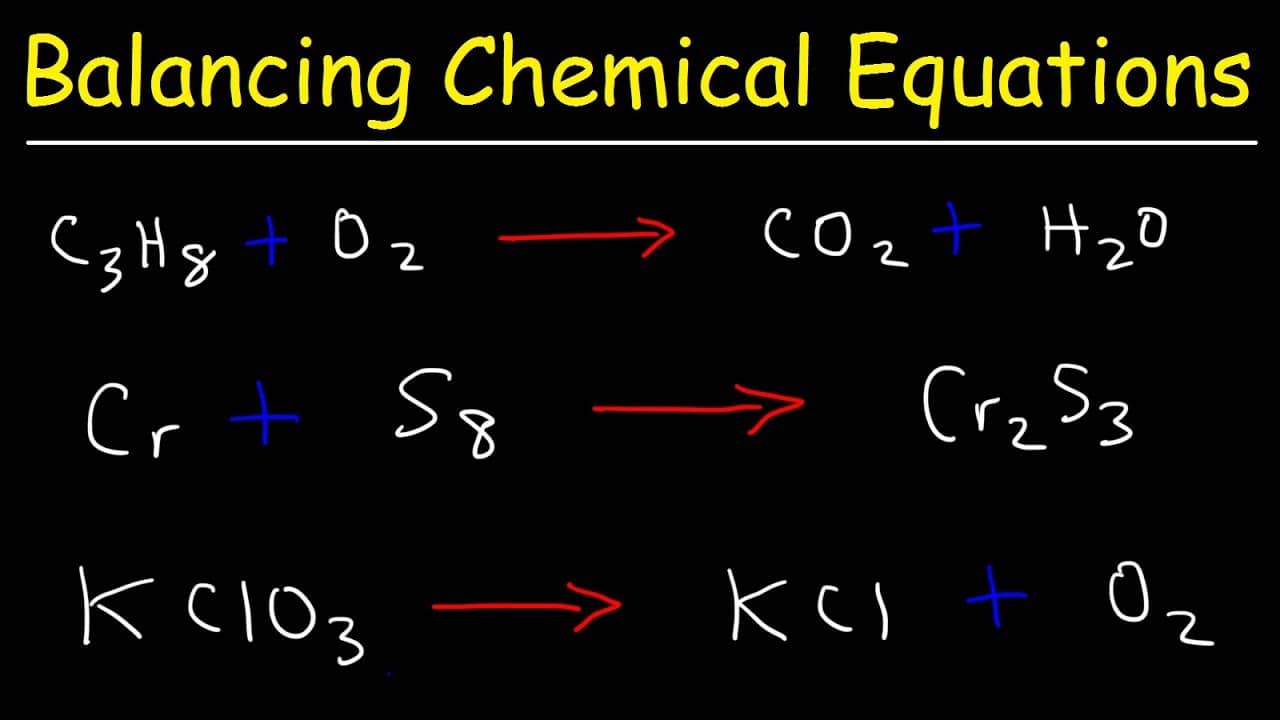
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
